The cold way is saccharification of grain according to gost. The number and methods of using enzymes for home brewing
Even beginners understand that carbohydrates are needed to get alcohol at home. Ideally, simple sugars: sucrose, glucose or fructose. In cereals, carbohydrates are present in sufficient quantities, but in the form of starch. Each starch molecule in turn consists of glucose fragments. When grain is taken as a raw material, before preparing the mash, starch is saccharified in it: it is divided into glucose molecules, only then the fermentation process becomes possible. Saccharification of starch in cereals can be accomplished by germinating part of the grains to produce malt. When germinated, enzymes are formed that break down starch into simple sugars.
The use of grain (malt) for the preparation of mash greatly ennobles the final drink. Grain moonshine is more soft than regular sugar.
Malt can be replaced with enzymes - Amilosubtilin and Glukavamorin. The role of the first is to break down starch molecules into smaller fragments, and the second is responsible for the processing of these fragments into simple sugars.
The recipe for cold mash on enzymes is much simpler than malt technology and is cheaper.
It is necessary to prepare:
- 3 kg of any raw material (any cereals, starch, flour, etc.);
- 10 l of water at room temperature;
- 12 g of Amylosubtilin and Glucavamorin;
- 75 g of fresh yeast.
The fermentation tank must be taken large, given the possible foaming. At least a third should remain empty.
Enzyme Mash Recipe
Cooking Mash:
- Bring the water to a boil, with constant stirring in small portions, add flour (cereal) and turn off the heat.
- When the mash cools down to a temperature of 80 ° C, add enzyme A to it and mix thoroughly.
- Leave to cool to a temperature of 65 degrees.
- At a mash temperature of 65 ° C, add enzyme D and mix thoroughly.
- Cover the pan and leave for 3-4 hours to saccharify the starch.
- Then pour the mash at room temperature into the fermentation tank, add activated yeast, close the lid, install a water seal and remove the container in a warm dark place.
- Estimated fermentation time is 7-10 days.
Enzymes provoke a rapid onset of fermentation, literally after 1-2 hours, bubbles will already be noticeable. The entire duration of the fermentation depends on the selected raw materials. It can range from 1 to 3 weeks. When using the recipe for mash on enzymes at home, it is important to track the readiness of the mash in time so that there is no souring. If a thin film appears on the brag, visible to the naked eye - it is urgent to start distillation. Distill the mash is best twice.
Before distillation, it is advisable to lighten the mash. This can be done using bentonite or simply set it for a day in the cold.
You can independently conduct saccharification with malt. This is the process of splitting potatoes, cereals or flour and other raw materials that contain starch under the action of natural enzymes. Sometimes artificial ingredients are used, which requires less effort. Which method of saccharification to choose is decided individually in each case.
What is the saccharification process for?
Cold or hot saccharification is necessary to make alcohol. Yeast alone is not enough. Sugar is important. It is found in cereals in the form of starch. This is a polysaccharide, which contains sucrose, fructose and glucose. Since only monosaccharides are needed to feed the yeast, the starch chain should be broken down into molecules before laying. If this is not done, then fermentation will not work.
Braga on enzymes of natural origin is performed hot. And if synthetic enzymes are used, cold saccharification is used.
Selection of ingredients and proportions
For hot saccharification take 4-5 liters of water per 1 kg of flour, cereal or other raw materials. Malt must be crushed and added at the rate of 150 g per 1 kg of feedstock.
To perform saccharification in a cold way, 1 liter of water is taken per 1 kg of raw material. Enzymes for are required in a volume of 5 g per 1 kg of raw material. Yeast will require 25 g of pressed type or 5 g of dry per 1 kg of feedstock, regardless of whether saccharification of flour, starch or any cereal needs to be performed.
Some recipes suggest the addition of other ingredients to the mash:
- antibiotics designed to prevent souring;
- feeding the yeast so that the fermentation process is faster;
- acid that stabilizes the acidity of the wort;
- defoamer.
Cold processing
Cold saccharification by enzymes is not done with malt. The natural ingredient is replaced with synthetic analogues. Glucavamorin processes starch to a sugar state, and amylosubtilin provides partial breakdown of molecules.
The technology is less expensive, easier compared to malt cooking, and the effect is not much different. Enzymes with water are added to the raw materials at the time of manufacture of the mash. Starch is transformed into sugar at about the same time as the fermentation process takes place.
Enzyme grain mash - cold saccharification - is a solution for those who are just starting to make alcohol at home, who do not have special equipment.
Cold processing does not require high temperatures and pauses. At the same time, the mash is cooked easier and faster.
The disadvantages of the technology include:
- the need to purchase enzymes;
- the importance of increasing the fermentation time to 10-20 days;
- the unnatural nature of the enzymes, which may leave a taste after several distillations.
Cold processing takes place according to the following technology:
- Add flour, starch, pasta or cereal to the container for the fermentation process, add water at a temperature of 35 ° C, supplement with enzymes and fill the yeast. To prevent increased foaming, the capacity is not filled by more than 70%.
- The mixture is closed with a water seal and rearranged in the dark, in a place where the temperature is not higher than 28 ° C.
- The fermentation process begins after 1 or 5 hours. In the first 2 days, fermentation is active, then the intensity is less. The process takes a week or 25 days.
- It is worthwhile to ensure that a thin film does not appear on the surface of the mixture. This suggests that the souring process has begun. In this case, the brew is urgently distilled.
- Ready mash is removed from the precipitate, distilled.
Hot processing
Hot saccharification is the traditional way. Grain sprouts in humid conditions, which starts the process of activating the necessary enzymes needed for the processing of starch. A grain that has sprouted to a suitable state is called malt. It can be of 2 varieties: light and green.
Green malt is used for saccharification of raw materials when 3 cm sprouts appeared. This product is stored for no more than 3 days. If you dry the cereal that has sprouted, it will already be light malt. It is stored longer. Both varieties of malt are quite effective.
Disadvantages of technology:
- a temperature is needed at which there is a risk that the raw material will burn;
- it is important to ensure temperatures up to 72 ° C for several hours, which is not always easy to create at home;
- sugared wort can sour quickly.
Hot saccharification by malt is carried out according to the following technology:
- Flour or cereal is poured with water at temperatures up to 50 ° C. It is necessary to stir the raw materials so that lumps do not turn out. 5 l of water are taken per 1 kg of raw material. Dishes should be filled to 75% and not more than this volume.
- The temperature is raised to 60 ° C, in this state is kept for 15 minutes.
- The mixture is brought to a boil and cooked for 1 or 2 hours, depending on the recipe. Cereals need to cook longer than flour. You should get a mushy mass of a homogeneous consistency.
- The composition is cooled to 63 ° C, malt is added to the mixture and does not stop stirring. For 1 kg of raw materials, 150 g of crushed malt is required.
- When the mixture reaches a temperature of 65 ° C, cover it and wrap it up to provide heat for 4 hours. Half the time indicated should be stirred every 30 minutes.
- To prevent souring, reduce the temperature to 25 ° C. Then add 5 g of dried or 25 g of compressed yeast per 1 kg of raw material. Then put a water seal, sent for fermentation in a dark place for a period of 2 to 6 days.
If you do not comply with the desired temperature, then saccharification will not work or it will be insufficient. Additional heating will not give the desired effect, since the enzymes cease to be active.
The process of saccharification with malt is only a step to get alcohol at home. When using natural ingredients and applying hot processing there is a risk of unnecessary trouble. But if you choose the right ingredients, observe the temperature regime and spend time on distillation of the alcoholic beverage, the result will be excellent.
Ethanol Production
The global ethanol market is about 4 billion decalitres (decalitres of absolute alcohol) per year. The leaders in ethanol production are the USA, Brazil, China. In the United States there are 97 plants for the production of ethanol from corn (another 35 plants are under construction) with a total capacity of 1.5 billion decalitres per year.
The main areas of ethanol use in world practice:
- 60% - additive to motor fuel;
- 25% - chemical industry;
- 15% - food industry (its share is declining).
Ethanol-based automotive fuel contains 10% ethanol (E-10 fuel) or 85% ethanol (E 85). With an oil cost of $ 60-70 per barrel, bioethanol becomes a competitive fuel. The introduction of ethanol in gasoline allows you to abandon the addition of tetraethyl lead to the fuel, resulting in reduced exhaust gas toxicity and fuel consumption.
In the United States, large-scale research is being conducted on the production of bioethanol from renewable plant materials (from stalks of corn, reed, etc.)
Under industrial conditions, ethanol is obtained by hydration of ethylene in the presence of a catalyst (H 3 PO 4 on silica gel), from hydrolysates of plant materials (wood, stalks of corn, cane), as well as from starch-containing raw materials (wheat, rye, triticale, potatoes), molasses, milk serum, Jerusalem artichoke. The average yield of 95.5% ethyl alcohol from 1 ton of various types of raw materials is presented in table 2.1.
Table 2.1
Ethanol yield from various types of raw materials
The end of table 2.1
At the distilleries of the Republic of Belarus (there are about 70 distilleries with a total capacity of more than 9 million decalitres per year), starch-containing raw materials are used for the production of ethanol, mainly cereal grain. The starch content in various types of grain is (in%): wheat - 48–57; rye - 46–53; barley - 43–55; oats - 34–40; millet - 42-60; corn - 61–70. Grain also contains (on average) ~ 3% sugar; fiber ~ 6%; pentosans and pectin substances ~ 9%; nitrogenous (protein) substances ~ 11%, fat ~ 3%.
Ethanol producers
In microbiological synthesis, the classic ethanol producers are yeast - saccharomycetes and schizosaccharomycetes. Most often use yeast Saccharomyces cerevisiae, Saccharomyces vini, Schizosaccharomyces pombe.
Saccharomycetes have cells of a rounded shape with a size of 10-15 microns, multiply by budding. Schizosaccharomycetes have large rod-shaped cells with a diameter of 4-5 microns and a length of 18-20 microns, multiply by division. Both those and other yeasts ferment glucose, mannose, fructose, sucrose, maltose well, ferment galactose harder and do not ferment pentose sugars (xylose, arabinose).
The theoretical yield of ethanol from 100 kg of fermented glucose is 51.14 kg or 64.80 l (this produces 48.86 kg of CO 2). In practice, the yield of alcohol is 82-92% of theoretical due to the consumption of part of the substrate for the reproduction and growth of yeast and the formation of by-products.
The synthesis of ethanol in a yeast cell is carried out according to the following scheme:
By-products of alcoholic fermentation are glycerin, higher (fusel) alcohols, organic acids (acetic, pyruvic, lactic, succinic), aldehydes. During alcoholic fermentation, sugar (glucose) is spent on the formation of various substances in the following amounts: ethanol - 46-47%, carbon dioxide - 44-46%, yeast biomass - 1.8-4.0%, glycerol - 3-4%, higher alcohols - 0.3-0.7%, organic acids - 0.2-1.0%, aldehydes - 0.1-0.2%. When yeast is returned to fermentation many times, sugar consumption for biomass formation decreases, and the fermentation intensity even increases slightly.
The formation of glycerol during alcohol fermentation is explained by the fact that during the induction period (before the formation of acetic aldehyde) between two molecules of phosphoglycerol aldehyde under the action of the aldehydmutase enzyme with the participation of a water molecule, a dismutation reaction occurs. In this case, one molecule of phosphoglyceric aldehyde is restored, forming phosphoglycerol, and the other is oxidized to 3-phosphoglyceric acid. Phosphoglycerol is not involved in further reactions and, after elimination of phosphoric acid, is a by-product of alcoholic fermentation. 3-phosphoglyceric acid undergoes transformations via the EMF path to form acetic aldehyde. After the appearance of acetic aldehyde, a stationary period of fermentation begins, in which the oxidation of phosphoglyceric aldehyde to phosphoglyceric acid proceeds in a more complex way, with the addition of an inorganic phosphate (EMF path). In this regard, along with ethanol, a certain amount of glycerol always forms during fermentation.
When acetic aldehyde is bound with bisulfite, the fermentation process is directed towards the formation of glycerol:
C 6 H 12 O 6 ® CH 3 CHO + CO 2 + CH 2 OH-CHOH-CH 2 OH.
In an alkaline medium, the molecule of acetic aldehyde enters a redox reaction with the second molecule, forming ethanol and acetic acid. At the same time, glycerol is accumulating. In total, the process is expressed by the following equation:
2C 6 H 12 O 6 + H 2 O ® ® 2CH 2 OH-CHOH-CH 2 OH + C 2 H 5 OH + CH 3 COOH + 2CO 2.
These techniques are used for the industrial production of glycerin.
Higher alcohols are formed from amino acids (to a lesser extent from keto acids) contained in a fermentation medium as a result of successive reactions of deamination of amino acids, decarboxylation of the formed keto acids and reduction of aldehydes.
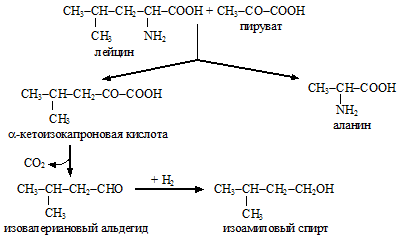
Of the higher alcohols in the mash there are: propyl (formed from threonine), isobutyl (from valine), amyl (from isoleucine) and isoamyl (from leucine).
 |
Currently, an intensive search is underway for non-traditional microorganisms producing ethanol that can ferment a wide range of substrates that have high ethanol productivity, have high resistance to ethanol and high temperature. Ethanol-synthesizing bacteria are of interest. For example, fish Zymomonas mobilis differ from yeast in intensive metabolism: they have a high specific rate of conversion of glucose to ethanol, provide a higher yield of ethanol (up to 95% of theoretically possible), are more tolerant to alcohol. But these bacteria are sensitive to the presence of inhibitors (furfural, phenols) in the nutrient media and require the fermentation process to be carried out under aseptic conditions.
Thermophilic bacteria Clostridium thermocellum (optimum growth temperature 68 ° С) they are capable of directly transforming the cellulose of plant materials into ethanol, but at the same time, the raw materials must be freed from lignin. It is not yet possible to achieve a high alcohol yield in the direct conversion of plant materials.
Yeast strains capable of fermenting pentose sugars ( Pachysolen tannophilus, Pichia stipitis, Candida shehata) The yield of ethanol during the fermentation of 100 kg of xylose reaches 35-47 liters.
In domestic practice, the production of ethanol from starch-containing raw materials using yeast Saccharomyces cerevisiaehaving an optimum fermentation temperature of 29–30 ° С.
Enzymatic starch saccharification
Traditional ethanol producers are incapable of breaking down polysaccharides, therefore, upon receipt of the wort, starch-containing raw materials must be digested and saccharified. The starch of most plants contains 20-25% amylose and 80-75% amylopectin. In plant cells, starch is in the form of grains (granules), the size of which ranges from 1 to 120 microns (potato starch has granules 40-50 microns in size, granules of grain starch - 10-15 microns). Starch, amylose and amylopectin are insoluble in cold water, alcohol, and ether. Amylose easily dissolves in warm water, amylopectin - when heated under pressure. The mesh structure of amylopectin molecules causes the starch granules to swell without dissolving (secondary bonds are weakened by hydration). At a certain temperature, the granules loosen, the bonds between the individual structural elements are broken, the integrity of the granules is violated. In this case, the viscosity of the solution increases sharply - starch gelatinization occurs. The paste is characterized by a random arrangement of molecules, a loss of crystalline structure. At a temperature of 120–130 ° С, the paste becomes easily mobile. The most complete dissolution of amylopectin occurs in wheat starch at 136–141 ° C, and in potato starch at 132 ° C.
Starch dissolved during the cooking of grain or potato is hydrolyzed (saccharified) by amylolytic enzymes of grain malt or cultures of microorganisms, mainly mycelial fungi and bacteria. Of the plant materials, the most abundant amylolytic enzymes are germinated cereal grains called malt. Currently, in the alcohol industry, enzyme preparations based on cultures of mycelial fungi (or bacteria of the genus Bacillus), which have several advantages over malt. Cultures of mycelial fungi are grown on wheat bran or corn flour, while conditional grains are required to produce malt. Extraneous microorganisms are introduced into the wort with malt in large quantities, which negatively affects the ethanol yield. The deep cultures of mushrooms are grown under sterile conditions; they do not contaminate the must with extraneous microorganisms. The cultivation of the surface culture of fungi is much faster (1.5-2.0 days) than the germination of grain (9-10 days). Mushrooms form a complex of enzymes that hydrolyze starch deeper and also break down hemicelluloses to monosaccharides, which increases the yield of ethanol from raw materials.
In the process of saccharification of starch-containing raw materials, various enzymes are involved. Of the greatest industrial importance are amylases. α- and β-amylases catalyze the cleavage of only α-1,4-glucosidic bonds. Under the influence of α-amylases, bonds break randomly, but mainly within the chains. As a result, mainly dextrins are formed, a small amount of maltose and oligosaccharides. Based on the nature of the action, α-amylase is called endogenous or dextrinogenous amylase.
The action of β-amylase is directed to the terminal (external) bonds in the starch; in this case, two glucose residues (maltose) are split off sequentially, starting from the non-reducing ends of the chains. β-amylase cannot bypass the branching site in the starch macromolecule; therefore, hydrolysis ceases at the penultimate α-1,4-glucosidic bond and high molecular weight dextrins remain during the hydrolysis of amylopectin. Amylose is almost completely converted by β-amylase to maltose, amylopectin - only by 50–55%.
As a result of the combined action of α- and β-amylases, a mixture of saccharides is formed, consisting of maltose, a small amount of glucose and low molecular weight dextrins, in which all α-1,6-glucosidic bonds of starch are concentrated.
In bacteria and microscopic fungi, β-amylase is absent, but it contains active α-amylase, which is distinguished by the composition of amino acids in the protein and the specificity of the action. In particular, when α-amylase catalyzes microscopic fungi, a large amount of glucose and maltose is formed. Among bacterial amylases, there are both sugar and dextrinogenous. The former hydrolyze starch by 60% and more, the latter by 30–40%. Microbial α-amylases, like malt α-and β-amylases, do not attack α-1,6-glucoside bonds.
Microscopic fungi contain glucoamylase, which catalyzes the cleavage of α-1,4- and α-1,6-glucosidic bonds in starch. When catalyzed by this enzyme, glucose residues are sequentially cleaved from the non-reducing ends of amylose and amylopectin. A water molecule joins at the site of bond breaking; therefore, the theoretical glucose yield during hydrolysis is 111.11% by weight of starch.
There are three possible ways of interacting an enzyme with a substrate (containing a large number of chains): multi-chain, single-chain and combined.
According to the multi-chain method, the enzyme molecule randomly attacks one of the polysaccharide chains, detaching a link from it, and then also randomly attacks the following chains, including, possibly, the one previously attacked. Thus, during the lifetime of the enzyme-substrate complex, only one catalytic event occurs.
In the single-chain method, the enzyme molecule, having randomly attacked one of the polysaccharide chains, sequentially cleaves the links from it until the chain is completely split. During the existence of the enzyme-substrate complex, all bonds available for the enzyme are hydrolyzed.
The combined method, or multiple attack method, consists in the fact that several bonds are hydrolyzed during the existence of the enzyme-substrate complex. Moreover, after cleavage of one link, the enzyme is not repelled, but delayed. The attack occurs with alternating single and multi-chain methods.
Studies have shown that α- and β-amylases carry out hydrolysis by the multiple-attack method (the multi-chain method is characteristic of bacterial α-amylase).
In domestic distilleries, raw (uns dried) malt in the form of malt milk, enzyme preparations (glucavamorin, amilorizin, amylosubtiline) of various levels of activity or a mixture of malt milk and an enzyme preparation are used to saccharify starch of raw materials.
The technology for producing malt includes the following main processes: soaking raw materials with a moisture content of 38–40%; germination of grain for 10 days in a pneumatic malt house in a layer 0.5–0.8 m thick; grinding malt in disk or hammer crushers; disinfection of malt with formalin or a solution of bleach and the preparation of malt milk. Malt milk is obtained by mixing crushed malt with water (4-5 l of water per 1 kg of malt).
Malt made from various cereal grains contains an unequal amount of each amylolytic enzyme. For example, barley malt has high α- and β-amylolytic activity, and millet malt is characterized by strong dextrinolytic activity. Most often, a mixture of three types of malt is prepared: barley (50%), millet (25%) and oat (25%). It is forbidden to use malt from one culture in the production of alcohol from the same culture.
If you decide to make moonshine, you need to choose a method of making mash.
There are many recipes for wheat mash, but only 3 technologies for the saccharification of starch-containing raw materials are the basis.
- Hot saccharification by enzymes or GOS
- Cold saccharification by enzymes or HOS
- Malt sugaring
The purpose of using enzymes is to prepare raw materials for fermentation with yeast. Pure starch yeast cannot be processed.
For its cleavage, the bacterial enzyme preparation Glucavamorin (Glucoamylase) is used. It works in tandem with amylosubtiline (alpha-amylase), which provides the preparation of raw materials for glucoamylase.
This is the main group of enzymes without which yeast will not consume starch. In addition to them, there are auxiliary enzymes such as Protosubtilin and Cellolux. They partially break down proteins and cellulose, increasing the yield of alcohol.
In malt, enzymes are produced during the germination of grain. For this, the grain is germinated to form a sprout 5-6mm. Then dried and sprouted sprouts and roots are removed.
Dosage of various enzymes
There are enough enzymes in the malt to sacrifice itself and another 4-5 kg. unmalted grain. Thus, for saccharification 1 kg. any grain requires 200-250g. Malt
The proportions of artificial enzymes depend on the shelf life and their activity, which is measured in units per gram.
You should know that enzymes are a catalyst for the process, and not a consumable unit. If you add less enzymes than required, the saccharification process will be delayed, but it will happen anyway.
For a specific calculation of the dosage of enzymes, you can use this calculator:
1. What enzymes are used and why?
In home distillation, enzymes came from industry. Their use in industry is due to a decrease in complexity, an increase in the stability of technological processes, an acceleration of the production process and an increase in the yield of alcohol in comparison with the use of traditional methods. Using a full complex of enzyme preparations allows you to get the maximum amount of alcohol from raw materials, as well as reduce the content of foreign components in the wort, which positively affects the organoleptic of the distillation product.
Modern industry uses enzyme preparations for thinning and saccharification of raw materials:
- Amilosubtilin GZh (AmiloLyuks, “A”) - for liquefying raw materials and preparing them for the action of other enzymes
- Glyukavamorin GZkh (GlyukaLyuks-A, "G") - for saccharification of starch
- CelloLux-A (“C”) - for saccharification of non-starchy polysaccharides (xylans, β-glucan, cellulose, pectins) or preparing them for the action of the above enzymes.
- Protosubtilin ("P") - for the breakdown of plant proteins, which leads to more active work of yeast
Thus, the minimum necessary enzymes during saccharification are Amilosubtilin and Glucavamorin. CelloLux-A and Protosubtilin perform additional saccharification and preparation for fermentation.
2. Dosage of various enzymes
Many questions cause the calculation of the dosage of enzyme preparations. Typically, the manufacturer or seller indicates the activity of dry enzymes in active units per gram of enzyme. There are also recommendations from the manufacturer on the dosage of active units of enzymes per gram of the substance being processed. Moreover, depending on those. the process, the number of enzymes can vary from minimum to maximum. Using this number, as well as using tables on the content of starch, protein and NPS (non-starchy polysaccharides), you can calculate the reference dose of each enzyme per kilogram of raw material.
The formula for calculating the number of enzymes per kilogram of raw materials is as follows:
Enzyme dose (gram) \u003d (P * R * 10) / A
- P - percentage of substance being processed (e.g. starch)
- R - recommended dosage of active units
- A - the activity of the drug in units per gram
It is worth adding that for some types of raw materials (rye) and enzymes with expired or near the end of their expiration dates, an increase in the dose of enzymes by 15-25% is required. Since there is practically no sense in calculating the exact dosage of drugs at home, you can make some simplifications in the calculation method by taking the maximum recommended values.
The table shows the calculation of the dosage of enzymes per 1 kg of raw materials:
Enzyme consumption in grams per 1 kg of raw material |
|||||||
| Raw materials | Starch | Protein | Cellulose | A-1500 units / g | G-3000 units / g | C-2000 u / g | P-120 u / g |
| Wheat | 56 | 16 | 6 | 0,75 | 1,16 | 0,90 | 4,38 |
| Barley (husked) | 49 | 13 | 7 | 0,65 | 1,01 | 1,05 | 3,79 |
| Corn | 68 | 7 | 3 | 0,91 | 1,41 | 0,45 | 2,04 |
| Rye | 50 | 15 | 2 | 0,67 | 1,03 | 0,30 | 4,38 |
| Triticale | 53 | 13 | 2 | 0,71 | 1,10 | 0,30 | 3,79 |
| Millet | 51 | 13 | 8 | 0,68 | 1,05 | 1,20 | 3,79 |
| Oats (peeled) | 37 | 13 | 10 | 0,49 | 0,76 | 1,50 | 3,79 |
| Potatoes | 18 | 2 | 2 | 0,24 | 0,37 | 0,30 | 0,58 |
| Rice | 73 | 8 | n / a | 0,97 | 1,51 | 2,33 | |
| Buckwheat | 64 | 12 | n / a | 0,85 | 1,32 | 3,50 | |
| Peas | 59 | 29 | n / a | 0,79 | 1,22 | 8,46 | |
Activity: | 1500 | 3000 | 3000 | 120 | |||
Unit consumption rate: | 2 | 6,2 | 30 | 3,5 | |||
Violations in the dosage of drugs to a lesser extent can affect the timing of the enzymes and the completeness of processing of raw materials. In this case, from a slight excess of the dosage, negative consequences were not noticed (except for overspending).
Thus, the universal recipe will be the use of 1 kg of raw materials:
- 1 gram - Amilosubtilin GZx 1500
- 1.5-2 grams - Glucavamorin GZx 3000
- 1 gram - CelloLux-A 2000
- 4-5 grams - Protosubtilin 120
3. Types of saccharification, their advantages and disadvantages
Nowadays, two different saccharification technologies are popular in home distillation - hot and cold, so named because of the different temperatures at which starch hydrolyzes. With hot saccharification, the raw material is heated to temperatures of 50-70 ° C and in this state is exposed to enzymes for 10-20 hours. At the same time, the risk of wort infection is minimal, enzymes act as efficiently as possible, but this method requires a lot of effort.
During cold saccharification using enzymes, the process proceeds at temperatures close to 30 ° C and with simultaneous fermentation. This method is less labor-intensive, but longer, and has a greater risk of souring the mash. The graphs show the dependence of enzyme activity on temperature over time:
| Amylosubtiline hydrolysis curves of grain batches at different temperatures (1 unit / g of starch) | Glucavamorin hydrolysis curves of grain batches at different temperatures (5 units / g of starch) |
The range of effective action of the enzyme Amilosubtilin corresponds to a pH range of 5.0-8.0 and a temperature of 50-75 ° C. For the enzyme Glucavamorin, the effective action lies in the following ranges: pH 3.0-6.5 and temperature 30-60 ° C.
It is worth adding that there are many intermediate methods between hot and cold saccharification, the use of which in many cases can be justified by specific conditions, the availability of components, time spent and other factors.
3.1 Hot Saccharification (GOS)
The recipe for mash using starch-containing raw materials and enzymes A and G:
- Prepare hot (boiling) water at the rate of ~ 6.5 liters of water per 1 kg of starch in raw materials (for cereals or crushed).
- Raw materials with constant stirring are added to hot water. For mixing, it is convenient to use a screwdriver or a low-speed drill with a nozzle for mixing building mixtures - “mixer”. Moreover, in order to avoid lumps, it is best to pour directly onto the nozzle rotating in the water.
- When the mixture cools to 75 ° C, a half dose of the amylosubtilin enzyme is introduced. Before making it can be dissolved with warm drinking water in a ratio of 1/10.
- Further, the wort is mixed from porridge to a liquid state or for about 30 minutes.
- The wort is allowed to cool to 56-58 ° C and the remainder of the enzyme Amilosubtilin and the enzyme Glucavamorin are introduced, then they are thoroughly mixed with a “mixer”. The enzyme working time at this stage will be about 1.5-2 hours.
- After the end of the saccharification process, the wort must be allowed to cool to a temperature of about 30 ° C. In order that the wort does not “become infected” during cooling, it is advisable to seal the container with it tightly.
- The wort is poured into a fermentation tank (previously sanitized), and yeast with a dosage of 2-3 grams of dry or 10-15 grams of pressed per kilogram of raw material is introduced into it. Fermentation takes place under a water trap.
The active phase of fermentation will last about 3-4 days, then the mash should be shaken periodically, without opening the fermentation tank.
3.2 Cold saccharification (ChOS)
The recipe for mash using starch-containing raw materials and enzymes A and G without brewing:
- It is advisable to crush the raw materials and be sure to clear the chaff, if any.
- Prepare water at a temperature of about 35 ° C at the rate of ~ 6.5 liters of water per 1 kilogram of starch in raw materials (for cereals or crushed). It is worth considering that it is undesirable to fill the fermentation tank with more than 7/10 volumes of braga.
- Half of the prepared water is poured into the fermentation tank.
- To reduce the likelihood of wort infection, it is recommended that you add an antibiotic, doxycycline (1 capsule per 20 liters of mash) to the water.
- Acidity is regulated within the range of 5-5.5 pH by phosphoric, sulfuric or citric acids.
- Next, the enzymes Amilosubtilin and Glucavamorin are introduced into the tank, according to the dosage per kilogram of starch in the raw material.
- If there is, then you can add defexilant defoamer - 1 ml per 20 liters of mash
- Raw materials are introduced, then everything is mixed.
- Yeast is added in accordance with the manufacturer's recommendations (10 grams of dry yeast per 4-5 liters of mash).
- The remainder of the water is added.
Fermentation occurs under a water lock with periodic stirring-shake (without violating the tightness). The fermentation process takes from one and a half to three weeks. The readiness for distillation is controlled by the appearance of a film on the surface of the mash. The appearance of the film is a sign that the mash is starting to turn sour and it needs to be distilled immediately. Ideally, the mash should be distilled shortly before the appearance of the film.